 And don’t forget to put the unit g/mol to your final calculated molar mass.First solve the brackets, then multiplications and at last do the final addition. Always follow the calculation order to avoid any mistakes in calculation.But all these units (i.e g/mol, grams/mole and g/mole) are the same. In some books, you may see the unit of molar mass as grams/mole or g/mole.I hope you have understood the short and simple calculation for finding the molar mass of Alum. Hence the Molar mass of Alum is 474.372 g/mol. Converting between Mass and Number of MolesĪ substance’s molar mass can be used to convert between the mass of the substance and the number of moles in that substance.Now, to calculate the molar mass of Alum, you just have to add the molar mass of all the individual atoms that are present in Alum. From the periodic table, the atomic mass of hydrogen is 1.008 g/mol, and the atomic mass of. Molar Mass Calculations – YouTube: This video shows how to calculate the molar mass for several compounds using their chemical formulas. Simply calculating the sum of the atomic mass of 2 H and 1 O will provide the molar mass of water. For example, since one atom of oxygen will combine with two atoms of hydrogen to create one molecule of water (\text is 58.44 g/mol. Chemistry is a physical science, and it is the study of the properties of and interactions between matter and energy. PLIX - Play, Learn, Interact and Xplore a concept with PLIX. Chemical Computations with Avogadro’s Number and the MoleĪvogadro’s number is fundamental to understanding both the makeup of molecules and their interactions and combinations. Simulations - Discover a new way of learning Physics using Real World Simulations. 
So, 1 mol contains 6.022×10 23 elementary entities of the substance. It is defined as the amount of a substance that contains as many particles as there are atoms in 12 grams of pure carbon-12. The mole (abbreviated mol) is the SI measure of quantity of a “chemical entity,” such as atoms, electrons, or protons. Periodic table with charges and groups Periodic table with charges and blocks The elements in blue colour are the s-block elements. The Periodic Table of the Elements 1 H Hydrogen 1.00794 2 He Helium 4.003 3 Li Lithium 6.941 4 Be Beryllium 9.012182 5 B Boron 10.811 6 C Carbon 12.0107 7 N Nitrogen 14.00674 8 O Oxygen 15.9994 9 F Fluorine 18.9984032 10 Ne Neon 20.1797 11 Na Sodium 22.989770 12 Mg Magnesium 24.3050 13 Al Aluminum 26.981538 14 Si Silicon 28. With Avogadro’s number, scientists can discuss and compare very large numbers, which is useful because substances in everyday quantities contain very large numbers of atoms and molecules. Visualize trends, 3D orbitals, isotopes, and mix compounds. Avogadro’s number is 6.022×10 23 molecules. Interactive periodic table showing names, electrons, and oxidation states. It is equal to 6.022×10 23 mol -1 and is expressed as the symbol N A.Īvogadro’s number is a similar concept to that of a dozen or a gross. Avogadro’s number is defined as the number of elementary particles (molecules, atoms, compounds, etc.) per mole of a substance. Although he did not determine the exact proportion, he is credited for the idea.Īvogadro’s number is a proportion that relates molar mass on an atomic scale to physical mass on a human scale. Avogadro’s Number Amedeo Avogadro: Amedeo Avogadro is credited with the idea that the number of entities (usually atoms or molecules) in a substance is proportional to its physical mass.Īmadeo Avogadro first proposed that the volume of a gas at a given pressure and temperature is proportional to the number of atoms or molecules, The solution is the concept of the mole, which is very important in quantitative chemistry. They also need a way to compare these numbers and relate them to the weights of the substances, which they can measure and observe. 
It is impractical to try to count or visualize all these atoms, but scientists need some way to refer to the entire quantity. The chemical changes observed in any reaction involve the rearrangement of billions of atoms. mole: The amount of substance of a system that contains as many elementary entities as there are atoms in 12 g of carbon-12. 
For example, the mean molecular weight of water is 18.015 atomic mass units (amu), so one mole of water weight 18.015 grams. The mass of one mole of a substance is equal to that substance’s molecular weight.This can also be written as 6.022×10 23 mol -1. 1.0 Li lithium 6.94 4 2+ 1.5 Be beryllium 9.01 Atomic number Electronegativity Molar mass (g/mol) Ion charge Atomic Symbol SOLID Name 5 2.0 B boron 10.81 6 2.5 C carbon 12.01 7 3- 3.0 N nitrogen 14.01 8 2- 3.5. 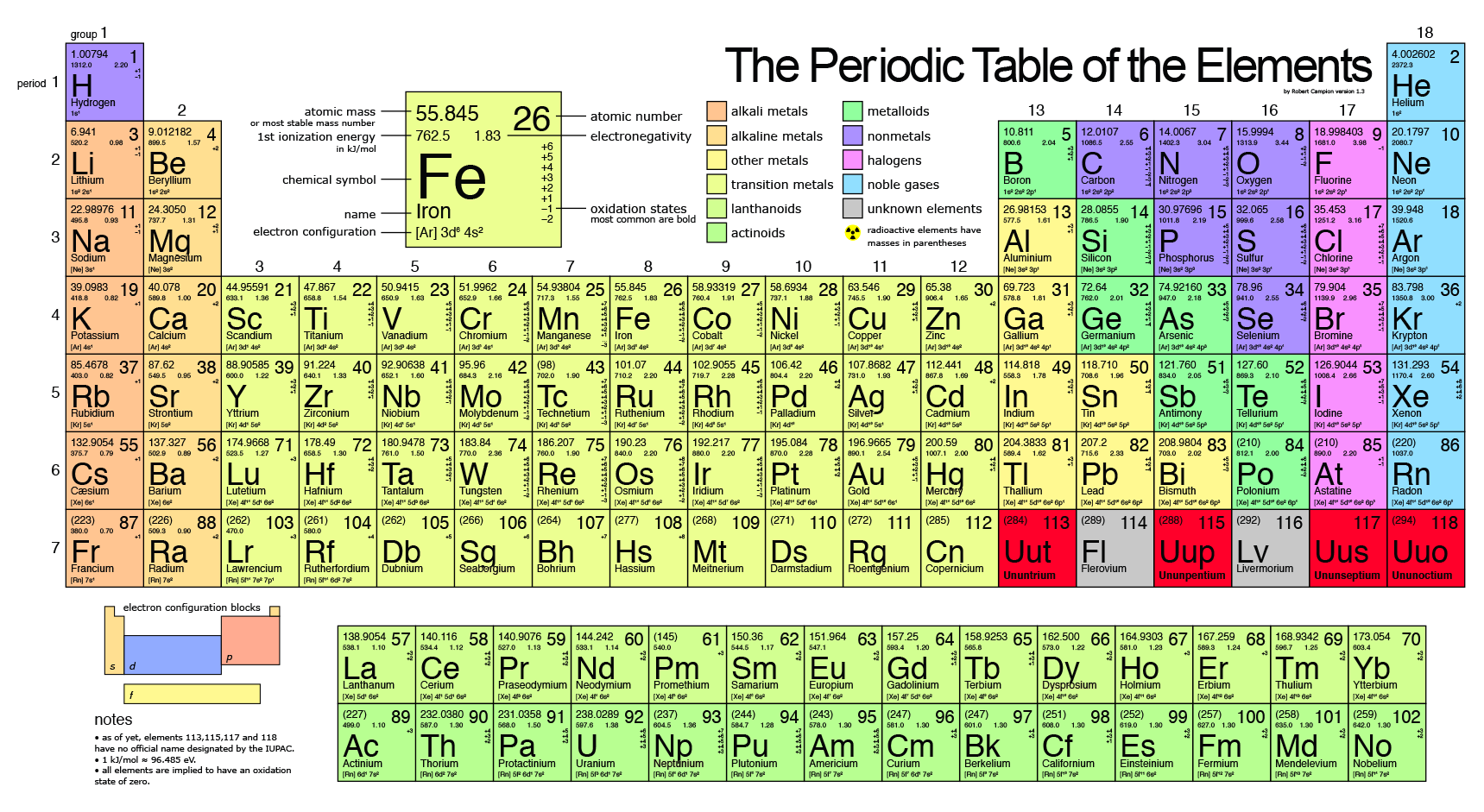
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
